BMS INTRACORONARY TEMPERATURE (ICT) CATHETER SYSTEM
Radius delivers miniaturized catheter technology engineered for complete simplicity.Designed to differentiate, the resulting IntraCoronary Temperature Catheter System Radius delivered to market optimizes safety and security, simplifies catheterization, and stands alone in the capabilities it provides.
Patient-Conscious, User Focused
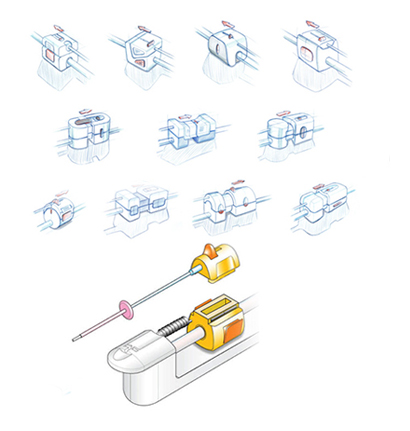
Radius was asked to create multiple components within a system that enabled user interaction to support the technology. Working with a global team of clinical stakeholders, Radius designers developed a range of subsystems that included a triggered catheter pullback system, a disposable catheter interface, and a sterile bag.
New Functionality Features
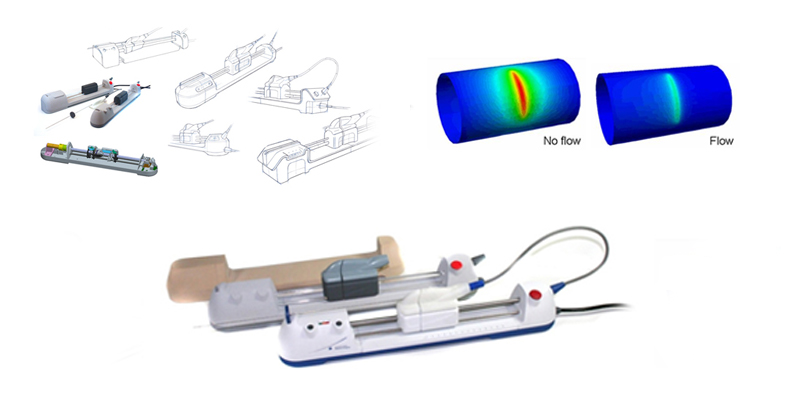
User insight led the team to develop a mobilized cart with CPU power and advanced, intuitive user interface, testing the concepts to gain ergonomic and functional understanding. Refinements were made to meet physical efficacy, security, and sterilization requirements and to run through Radius’ robust quality management system to ensure the resulting system and documentation were ready for clinical trials.
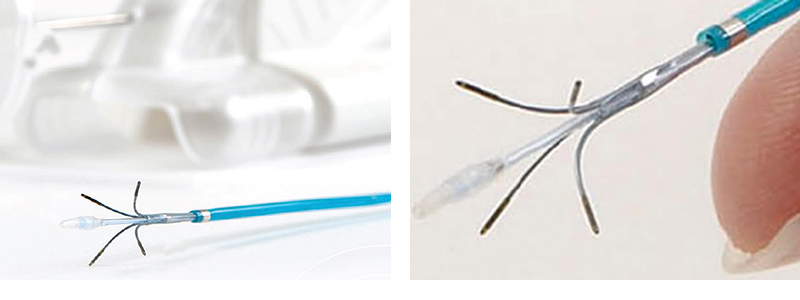
A simplified user interface added new automated security triggers for more procedural security and included temperature-sensor screens for robust thermal imaging that enables doctors to investigate previously undetectable regions of the heart.
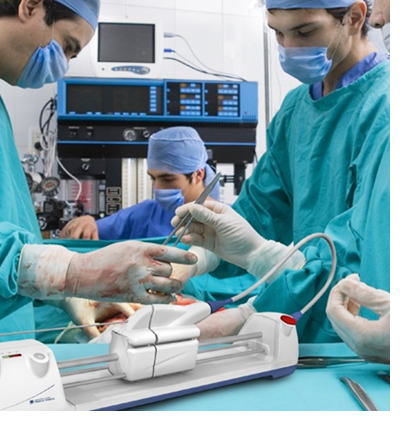
The IntraCoronary Temperature Catheter System, executed within a tight schedule and with an eye toward manufacturing production, offered Bristol-Myers Squibb a fully integrated quality management system (QMS) within FDA timelines. Inspired in design, sophisticated in its approach, the system maximizes safety features and turns a complex procedure into its simplest, most robust form.
Innovation. Made Possible. Made Better.
Visit Bristol-Myers Squibb.
RELATED CONTENT
RELATED INSIGHTS
RELATED CAPABILITIES
WANT TO LEARN MORE?
Start a conversation with Radius to learn how we can help you take your Healthcare project to the next level.